“Technology is the closest thing to magic we have in the real world, and Global Good wields its inventive magic to change the lives of those who need it most—the billion-plus who struggle on the bottom rungs of the economic ladder. We’re always looking for ways to bring our ideas to market faster and at a lower cost. Carbon is helping us do that.”
Nathan MyhrvoldFounder, Chief Executive Officer, Intellectual Ventures
The Global Good Fund, a collaboration between Intellectual Ventures (IV) and Bill Gates to develop technologies for humanitarian impact, aims to address this gap by developing an easy-to-use and affordable early tuberculosis diagnostic device for countries with limited healthcare infrastructure. To create the technology, they partnered with Carbon to design, engineer, and manufacture a device that could be used in field trials (Figure 1). This case study explains how Carbon’s technology helped expedite the diagnostic cassette product development cycles and time to market to help save lives globally.

Figure 1: Tuberculosis diagnostic cassette, 3D manufactured using Carbon technology
THE CHALLENGES
Global Good, together with the scientists at IV Lab, work in multiple research areas, including focusing on the very beginning stages of invention, validating a concept, and refining the technology to demonstrate its potential to make an impact around the globe. Driven by this mission, they wanted to develop a new accurate TB diagnostic method that was fast, easy to use, and lower cost.
Traditional methods of diagnostics are expensive and slow, and thus not ideal for low-resource settings where the majority of all new TB cases occur.1 Another challenge is that sputum collection, which is critical to the TB diagnostic process, is difficult for the elderly, children, and HIV patients and poses a risk to healthcare workers because the process can produce pathogen-carrying aerosols.
PRODUCT DEVELOPMENT—NEW DESIGN APPROACH
The team, inspired by simple-to-use lateral flow assay (LFA) tests already developed for diagnosing malaria and for in-home pregnancy tests, set out to develop a product that solves two of the three challenges—speed and usability—using a new product design. Instead of requiring a sputum sample, the device would use urine and detect a biomarker for TB. The proposed TB test would also contain a lateral flow strip with detection reagents encapsulated inside a plastic cassette (Figures 1 and 2). With the initial design proposal addressing the fast detection and sputum-free challenges, the next challenge was to design, engineer, and manufacture this plastic cassette cost-effectively for field evaluations.

Figure 2: A lateral flow strip is placed between the two 3D printed parts of the diagnostic cassette housing shown above
PRODUCT DEVELOPMENT—ACHIEVING LOWER COSTS AT HIGH SPEED
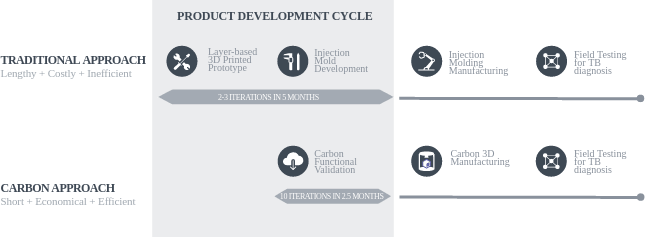
With any new product development, design and prototyping successes in the early stage are a challenge. The IV Lab product development team not only embraced a new design approach but also achieved significantly lower product development costs at a much higher speed of innovation. Previously, the product development cycle at IV Lab had four distinct phases: (1) design and prototyping, (2) mold development, (3) injection molding product manufacturing, and (4) field testing (Figure 3). The IV Lab team began with a layer deposition 3D printing technology, such as Fused Deposition Modeling (FDM), to create prototypes, followed by short-run injection molding of the final part.
Figure 3: Comparing cassette product development approaches: Traditional versus Carbon
However, the team quickly ran into problems with the traditional approach because prototyping the cassette with existing layer-based technologies is slow, and injection mold iterations are lengthy and expensive—typically, $25,000 USD per mold. As a result, the IV Lab team was able to complete only two to three prototyping iterations, followed by two to three injection mold development iterations, over the course of approximately five months of the product development cycle (Figure 3). The team encountered numerous mold failures and mold modification-related delays. These mold costs added significantly to the fixed cost of product development and created an economic barrier to carrying out product iterations, not ideal when the mission was to redesign a new cost-effective product for low-resource countries. Frustrated with the cost and the slow product development process, the IV Lab team decided to seek a more efficient method.
IV Lab discovered a better way to manufacture when their product development team was introduced to Carbon’s M Series printer powered by Digital Light Synthesis™ technology. Working in close partnerships with Carbon engineers, IV Lab 3D printed the first cassette design iteration in less than a week using Carbon’s Rigid Polyurethane (RPU) 70 material. Carbon’s easy-to-use software and industrial printer design helped IV Lab engineers come up to speed quickly and start iterating on the product design immediately. Carbon’s end-to-end digital manufacturing process enabled IV Lab engineers to carry out 10 product design iterations in half the amount of time compared to the traditional approach (Figure 3). Moreover, with 3D Manufacturing there is minimal material waste, no constraints resulting from minimum order quantities of parts, and no maintenance of parts inventory as a result of the flexibility of on-demand manufacturing.
Another significant advantage of Carbon’s manufacturing technology is that IV Lab could perform product iterations, functional validation, and actual manufacturing on the same machines and with the same materials throughout the entire product development cycle. Traditional manufacturing requires different materials and different technologies at each stage, which results in reduced flexibility, long delays, and operational inefficiencies. Leveraging Carbon’s technology, IV Lab was able to reduce end-product costs significantly while increasing the number of product design iterations by two to three times. The cost reduction is a phenomenal achievement for IV Lab given the mission to create a low-cost solution for the developing world.
CONCLUSION
3D Manufacturing is a transformative journey, and the IV Lab product development team successfully took the leap from slow prototyping and inefficient traditional manufacturing approaches to a fast, efficient 3D Manufacturing approach with Carbon. The IV Lab team successfully field tested more than 1,000 of these 3D manufactured TB diagnostic tests. In its mission to invent technology to solve some of humanity’s most daunting problems, Global Good and the IV Lab team leveraged Carbon’s technology to deliver a low-cost product that is easy to use, contributes to fast diagnosis of TB, and helps pave the path to eliminating the deadly disease.
If you are interested in learning more about how Carbon’s technology can help you create new disruptive and differentiated products, please contact the Carbon sales team.
¹http://www.who.int/tb/global-tb-report-infographic.pdf?ua=1